Paternò–Büchi reaction
Ìrísí

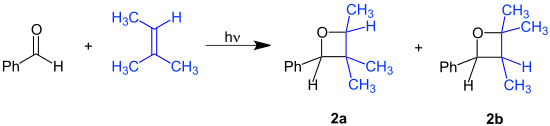
Paternò–Büchi reaction tí wọ́n sọ lórúkọ Emanuele Paternò àti George Büchi tí ó ṣẹ̀dá lílò àti ìrísí rẹ̀, jẹ́ àdàpọ̀ kẹ́míkà ṣiṣẹ́ tí ó maa ń di ẹgbẹ́ oxetane róbótó mẹ́rin lati carbonyl àti alkene.[1][2][3]
Pèlú èròjà benzaldehyde àti 2-methyl-2-butene àbáyọrí rẹ̀ ní àdàpọ̀ ìrị́sí àwọn isomer:
Àwọn èròjà míràn ni benzaldehyde àti furan[4]
Àwọn ìtọ́kasí
[àtúnṣe | àtúnṣe àmìọ̀rọ̀]- ↑ E. Paterno, G. Chieffi (1909). Gazz. Chim. Ital. 39: 341.
- ↑ G. Büchi, Charles G. Inman, and E. S. Lipinsky (1954). "Light-catalyzed Organic Reactions. I. The Reaction of Carbonyl Compounds with 2-Methyl-2-butene in the Presence of Ultraviolet Light". Journal of the American Chemical Society 76 (17): 4327–4331. doi:10.1021/ja01646a024.
- ↑ Thorsten Bach (1998). "Stereoselective Intermolecular [2 + 2]-Photocycloaddition Reactions and Their Application in Synthesis". Synthesis 1998: 683–703. doi:10.1055/s-1998-2054.
- ↑ Paternò–Büchi Reaction as a Demonstration of Chemical Kinetics and Synthetic Photochemistry Using a Light Emitting Diode Apparatus Matthew P. Thompson, Jonathan Agger, and Lu Shin Wong Journal of Chemical Education 2015 92 (10), 1716-1720 doi:10.1021/acs.jchemed.5b00129